Gene Editing Beyond CRISPR-Cas9: Base Editors
Can today’s gene editing technologies help? CRISPR-Cas9 is versatile but lacks efficiency when correcting single-base mutations. Newer tools, however, are more effective, minimizing double-strand breaks and eliminating the need for donor templates. Recently, determined researchers at Harvard University discovered base editors, arming the CRISPR toolbox with a new set of molecular machines (Ravindran, 2019.)
Construction of a Base Editor
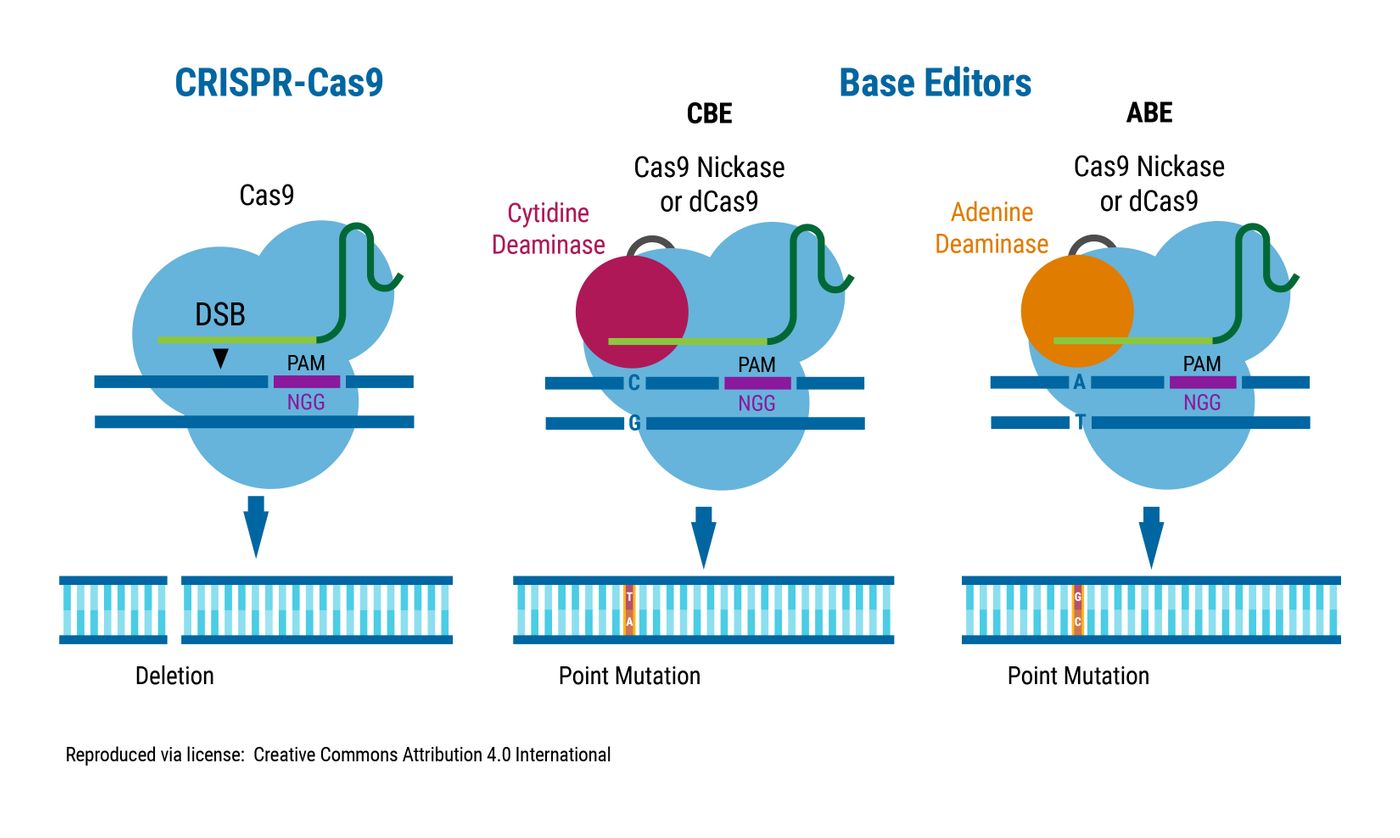
When either catalytically dead Cas9 or Cas9 nickase is fused to a nucleobase deaminase, guide RNA (gRNA) directs the base editor to a desired target sequence. Bases are swapped, and cells repair the DNA (Rees, H., 2018.)
Komor, et al. created the first Cytosine Base Editor using the naturally occurring cytidine deaminase, APOBEC1. When gRNA directs the fusion protein to a target site, the cell machinery converts C-to-T, and G-to-A on the opposite strand (Komor, 2016.)
An Adenine Base Editor (ABE) converting A-to-G was much harder to create. Gaudelli et al. persisted and harvested a tRNA adenine deaminase from E. coli. When target-bound, this ABE causes an A-to-G change and T-to-C in the opposite strand (Gaudelli, 2017.)
Application and Delivery of Base Editors
Researchers are using these molecular machines to correct and study pathogenic mutations and to generate animal models. The editing efficiency of base editors is almost 10-fold higher than standard CRISPR-Cas9 with less than 1% indel formation. Base editors can be delivered into primary cells, cell lines and even embryos as DNA, RNA or Ribonucleoprotein complexes. Microinjection or electroporation have been used for delivery (Anzalone, 2020.)
Limitations and Evolution of the Base Editor
Like CRISPR-Cas9, base editors can also produce off-target effects, the clinical relevance of which must be evaluated. Base editors operate within a five nucleotide “editing window.” Sequences outside this window will be edited less efficiently, but bystander mutations can still occur in nearby C’s and A’s. Recently, more evolved base editors with optimized targeting windows have been developed to reduce these undesired effects (Rees, 2018.)
A limitation of base editing concerns the genomic sites that can be targeted, as this is governed by the proximity to the PAM sequence recognized by the Cas9 component. Scientists are working to expand base editor applications by using Cas9 variants that recognize a broader range of PAM sequences.
The Future of Base Editing
David Liu likened CRISPR to a pair of molecular scissors that splice DNA, whereas base editors are more like a pencil and eraser (Gaudelli, 2017.) Easy to use and readily available from the non-profit plasmid repository, Addgene, soon, they could be used ex vivo to edit hematopoietic stem cells or T cells before administration to patients as a promising cell therapy.
Many companies are using base editors in their therapeutic pipelines. Dr. Liu co-founded Beam Therapeutics, Inc. to develop precision genetic medicines to cure hemoglobinopathies. In a recent publication, Dr. Gaudelli et al., used MaxCyte® electroporation to deliver mRNA encoding a second-generation ABE variant to edit the HBG1/2 promoter region. This resulted in higher upregulation of fetal gamma-globin than the first-generation ABE. The same highly functional editors were used for multiplexed gene editing in activated T cells to knock out the TRAC and B2M genes for use in allogeneic CAR therapies (Gaudelli, 2020.)
Until recently, precise gene editing was in the realm of science fiction. Now, it is a reality making it possible to edit the human genome, one base at a time.